Presenter
Jason K Cooper
Contributors
Zemin Zhang, Sebastian Reyes-Lillo, William Goddard III, Qi An, Elena Corbae, Sarah Lindley, Colin Ophus, Karen Bustillo, David Larson, Jason K. Cooper
Abstract
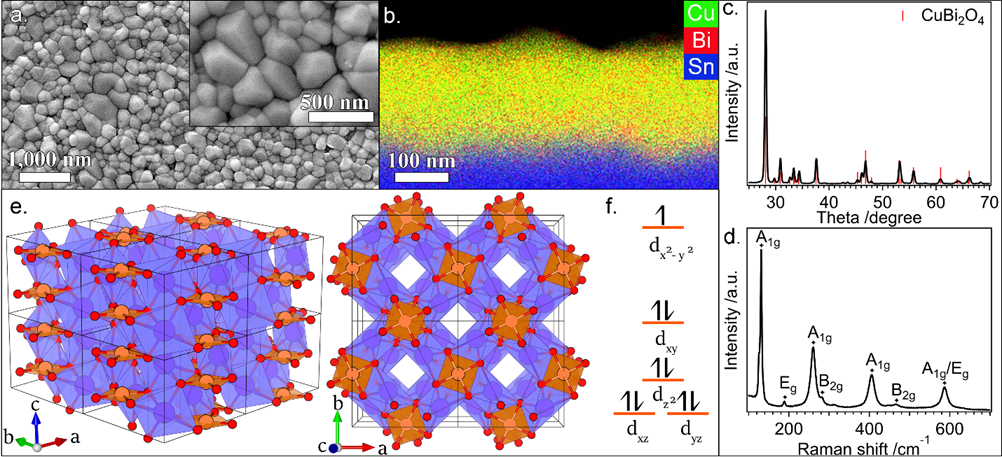
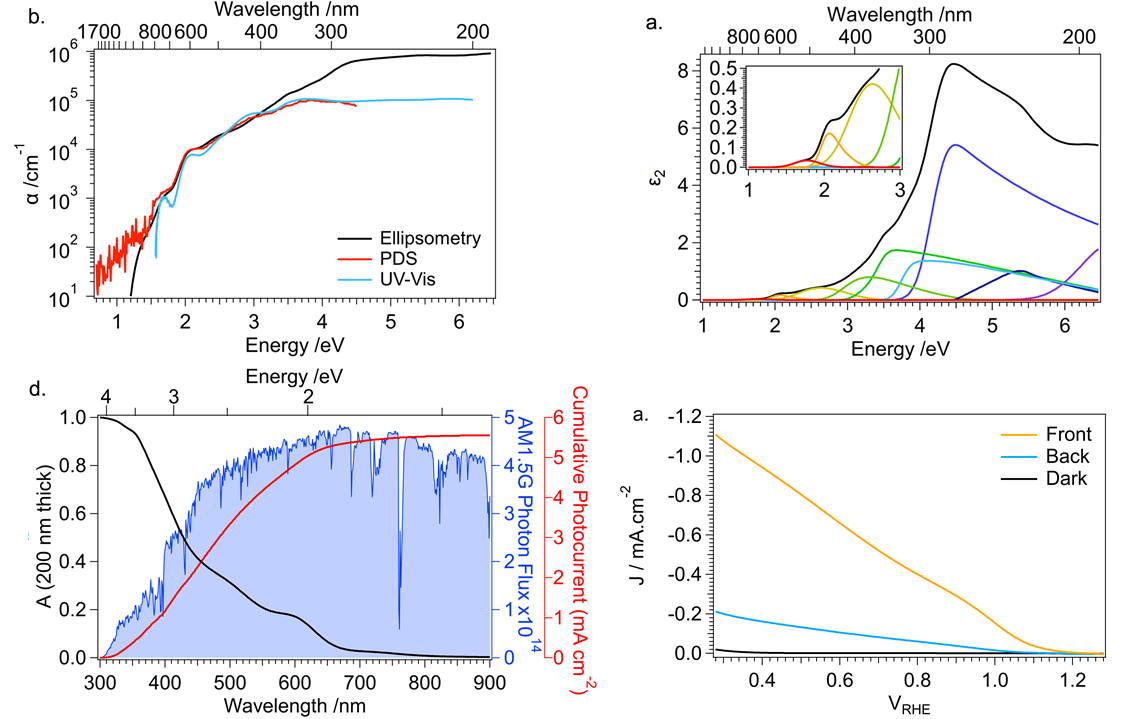
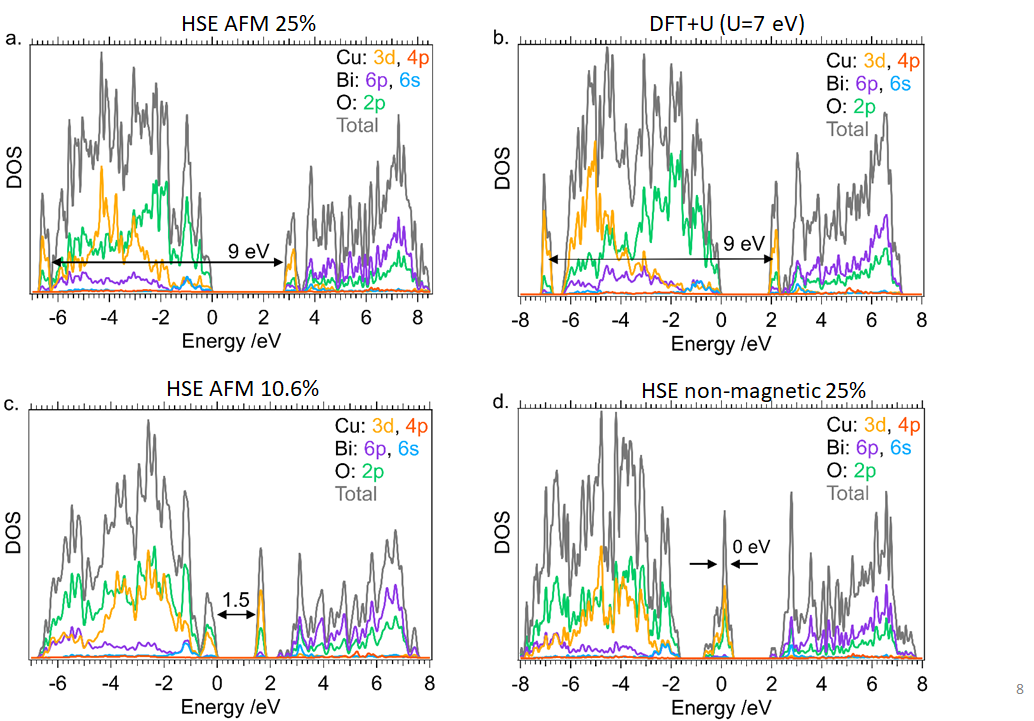
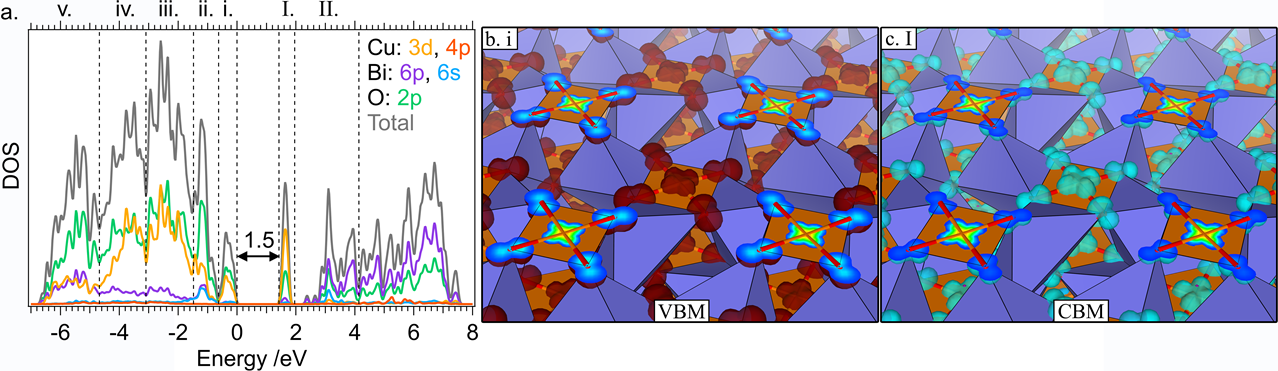
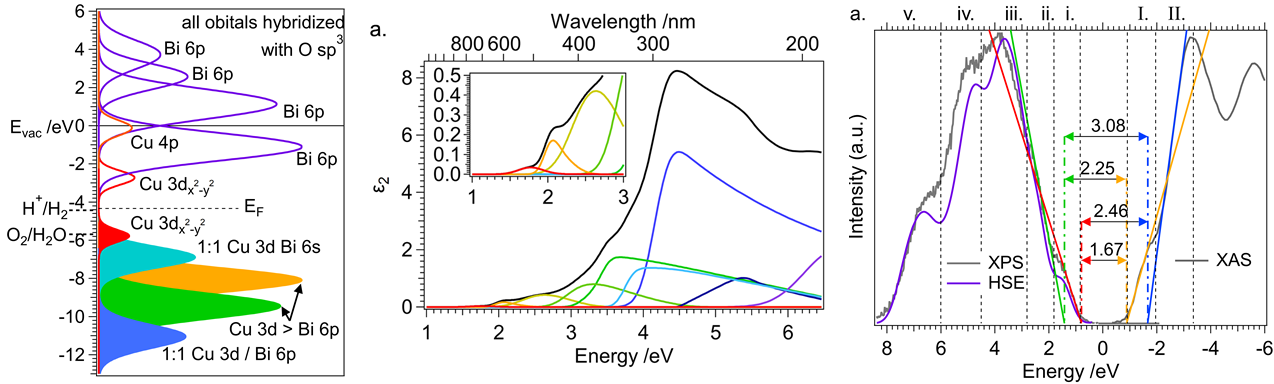
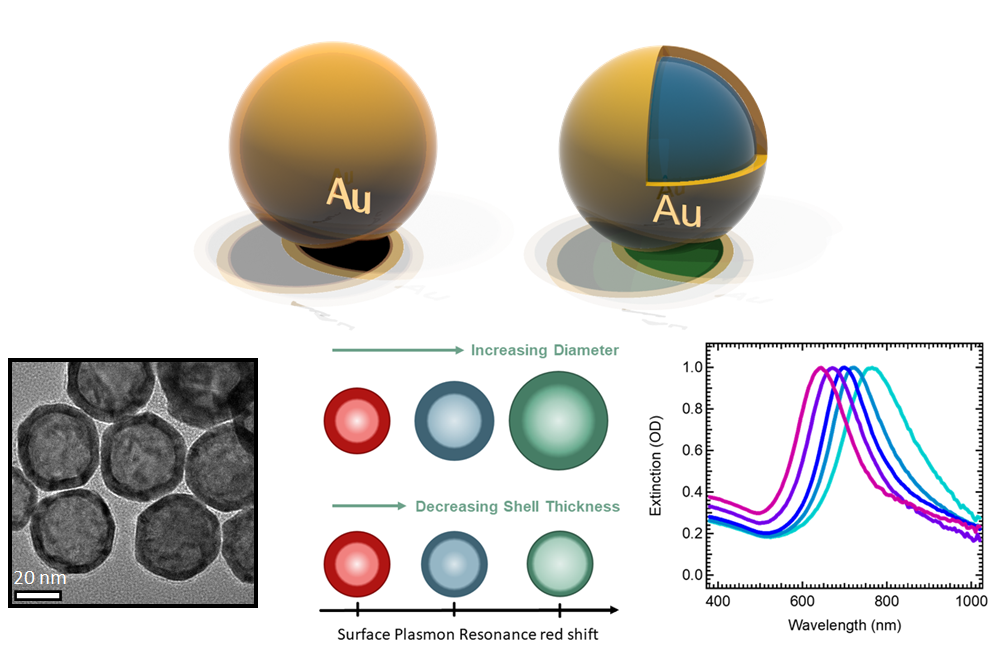
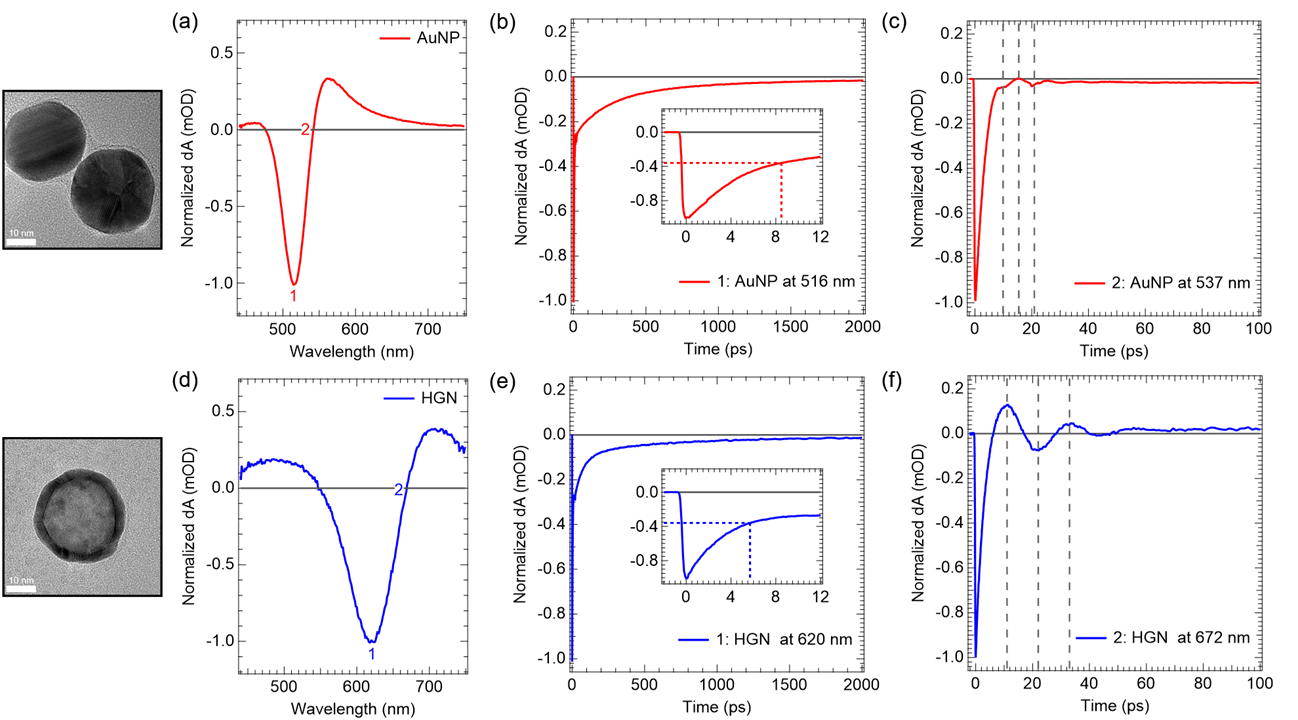
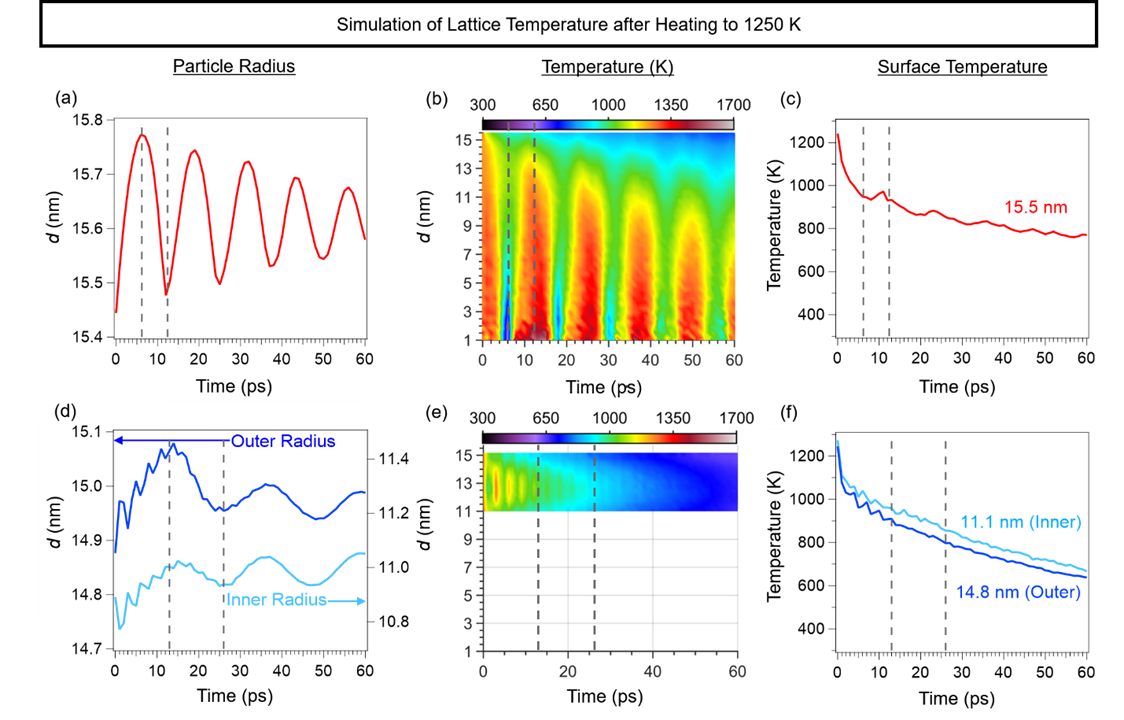
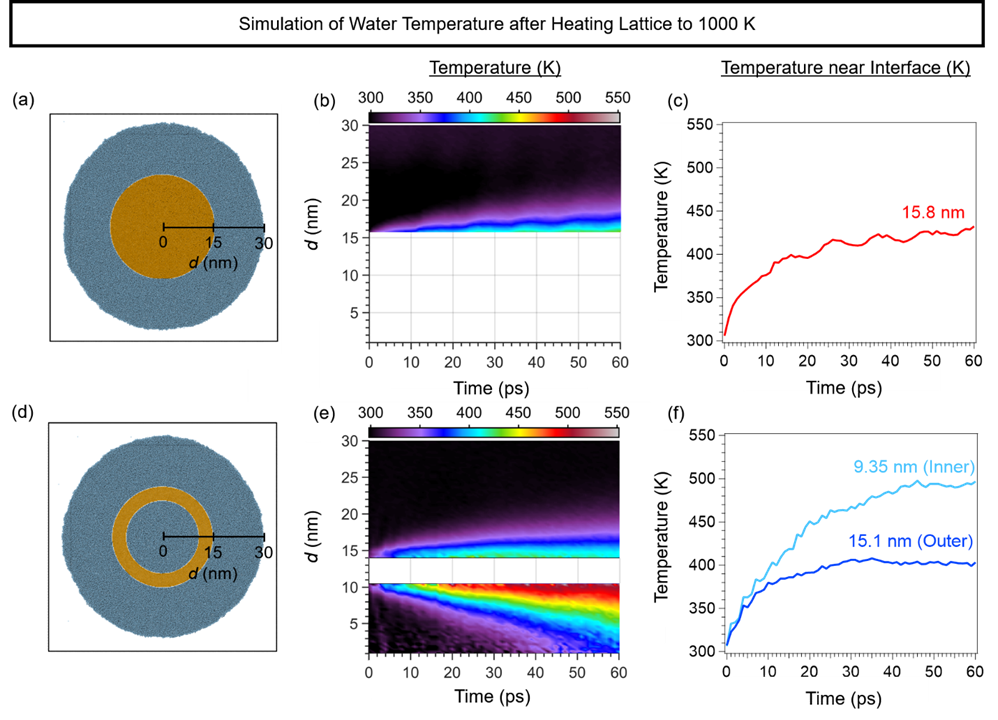
The economies of the future and the sustainability of the planet’s ecosystems will rely on green energy technologies such as through the capture of sunlight to drive chemical transformations. In particular, light-harvesting photoelectrodes dictate the generation of photovoltage and photocurrent so understanding the underlying physics governing performance and stability limitations are critical to advance related technologies. Specifically, CuBi2O4 is a natively p-type semiconductor with 1.5 – 1.8 eV optical bandgap; as a photocathode it has been demonstrated to have a photocurrent onset potential greater than 1 VRHE yet remains limited to about 1.5 mA cm-2. In this investigation we characterize the electronic structure (valence and conduction band orbital composition) of the material using a suite of advances spectroscopic techniques in conjunction with DFT modeling. These results suggest the limited electron polaron diffusion length of 45 nm is related to charge localization at Cu 3d states in the conduction band. Photoelectrodes made of plasmonic nanostructures are interesting for energy conversion applications due their dual function acting both as catalytic centers and light absorbers thus localizing the light energy directly at the catalytic site. Hollow-gold nanospheres (HGNs), thin shells of gold which can be readily dispersed in water, have a plasmon resonance absorption which can be tuned from 520 nm to the near infrared based on the shell dimeter and thickness making them excellent structures to harvest a large portion of the terrestrial solar flux. After the plasmon is excited, the energy is rapidly converted to heat the particle. In this study, through a combination of transient absorption (TA) spectroscopy and full scale molecular dynamics (MD) on 30 nm particles surrounded by water, the spatiotemporal temperature and pressure profiles reveal that photoexcitation of the HGN creates a high temperature and high-pressure environment particularly suited for thermal catalysis. These studies taken together identify an unique opportunity for coupled reaction micro-environments to take advantage of photoelectrochemical and photothermal reaction mechanisms.