2018: Applied Materials and Interfaces
https://doi.org/10.1021/acsami.8b02977
Chang-Ming Jiang, Gideon Segev, Lucas H. Hess, Guiji Liu, Gregory Zaborski, Francesca M. Toma, Jason K. Cooper, Ian D. Sharp*
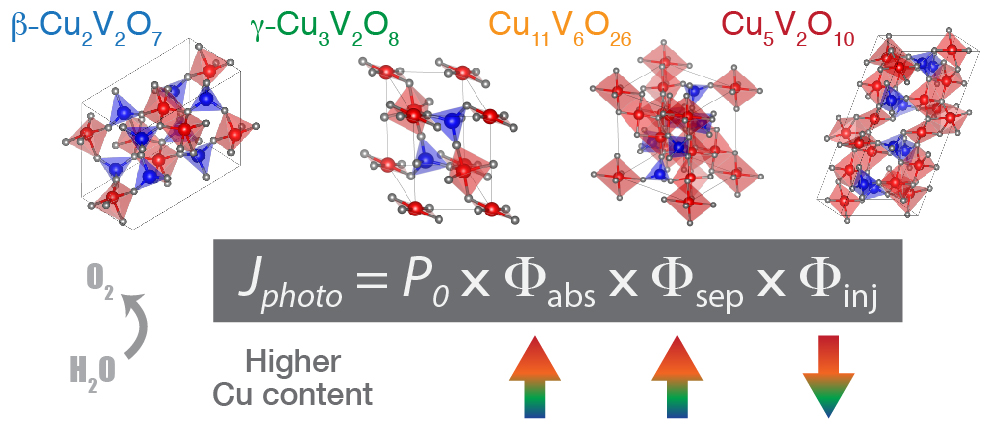
To understand functional roles of constituent elements in ternary metal oxide photoanodes, essential photoelectrochemical (PEC) properties are systematically analyzed on a series of copper vanadate compounds with different Cu:V elemental ratios. Homogeneous and highly continuous thin films of β-Cu2V2O7, γ-Cu3V2O8, Cu11V6O26, and Cu5V2O10 are grown via reactive co-sputtering and their performance characteristics for the light-driven oxygen evolution reaction are evaluated. All four compounds have similar bandgaps in the range of 1.83–2.03 eV, though Cu-rich phases exhibit stronger optical absorption and higher charge separation efficiencies. Transient photocurrent analysis reveals a reduction of surface catalytic activity with increasing Cu:V elemental ratio due to competitive charge recombination at Cu-related surface states. This comprehensive analysis of PEC functionalities—including photon absorption, carrier separation, and heterogeneous charge transfer—informs strategies for improving PEC activity in the copper vanadate materials system and provides insights that may aid discovery, design, and engineering of new photoelectrode materials.